Enhanced local and systemic inflammatory cytokine mRNA expression in women with endometriosis
Aug 10, 2020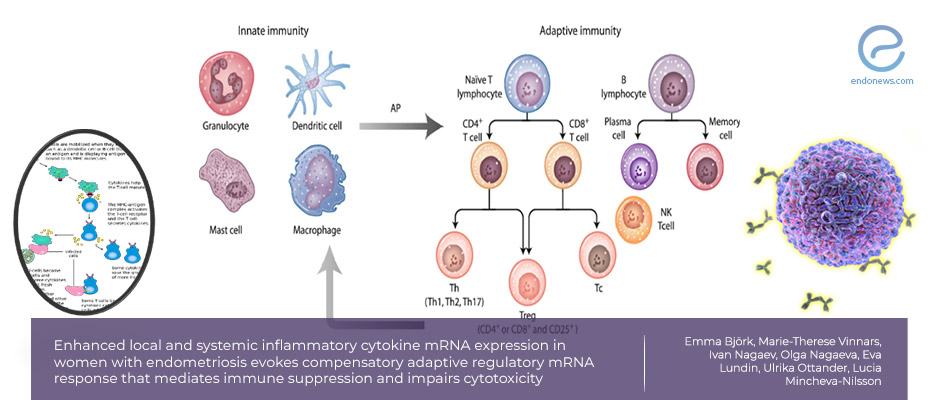
Regulatory T cells have the ability to control the cytokine secretion and suppress the cytotoxicity of the immune system in endometriosis.
Key Points
Highlight
- Immune mechanisms play an important role in the pathogenesis of endometriosis.
Importance
- The interplay and contribution of cytokines in the overall immune response in endometriosis are understood.
What is done here?
- Tissue from an endometriotic lesion, eutopic endometrium, and peripheral blood was collected from 31 patients with endometriosis (at surgery) and 18 healthy age-matched controls.
- mRNA for eleven cytokines, namely cytotoxic Th1 (IFNγ and IL15), humoral Th2 (IL 4), regulatory Tr1/Th3 (IL10 (Tr1) and/or TGFβ1 (Th3), and inflammatory (IL1β, IL6, IL8, tumor necrosis factor (TNF)α, and LTA/TNFβ) were evaluated and quantified on paired samples of endometriotic tissue, endometrium, and peripheral blood mononuclear cells from women with endometriosis and healthy controls.
- Quantitative Real-time PCR was used to characterize mRNA.
- Immunohistochemical staining (IHC) with monoclonal antibodies was performed to examine T regulatory cells CD4, CD25.
Key results
- High levels of mRNA expression of inflammatory cytokines, mainly IL-6 and IL8, IL1β, and (TNF)α resulted in an extensive systemic and local inflammatory response in endometriosis patients compared to healthy controls.
- An impaired upregulation of IFNγ, a key cytokine necessary for the Th1 response that mediates NK- and cytotoxic T-cell killing was detected.
- Upregulation of mRNA for IL-10 and IL-2 in the endometriosis patients, prominent in endometriotic lesions, consistent with a local Tr1 regulatory response priming naïve T cells into adaptive regulatory T cells (Treg cells) was observed.
- A substantial abundancy of CD4+CD25++ cells in the endometriotic lesions was detected as shown by IHC staining of serial tissue sections.
Limitation of the study
- The results could at least provide a mechanistic explanation of the pathogenesis of endometriosis but still are not enough to explain the etiology of the disease.
Lay Summary
Endometriosis is not only characterized by the implantation of endometriotic tissue outside of the uterine cavity but also necessitates an impaired immune response. It is suggested that abnormal immunological mechanisms causing the dysfunction of immune cells and their mediators are involved in the development of endometriosis. In the absence of appropriate immune control, the ectopically disseminated endometrium continues to lodge, proliferate, and invade the underlying tissue forming endometriotic lesions that grow under the hormonal influence of the menstrual cycle.
Cytokines may play a role in the present understanding of the pathogenesis of endometriosis. They modulate the patient’s immune system toward the permission for ectopic implantation of endometrial tissue resulting in the progression of this chronic disease. Cytokines are small proteins/peptides secreted by a variety of cells. Cytokines function as key mediators of intercellular communication.
The authors of the current study entitled "Enhanced local and systemic inflammatory cytokine mRNA expression in women with endometriosis evokes compensatory adaptive regulatory mRNA response that mediates immune suppression and impairs cytotoxicity", by Dr. Mincheva-Nilsson et al. from Sweden, recently published in the "American Journal of Reproductive Immunology", aimed to understand the role of cytokines in the overall immune response in endometriosis. Different cytokine profiles are associated with the immune system to regulate immunity, inflammation, promote or prevent cell growth, movement, and exert immune control.
The results of the study showed that the high levels of mRNA expression of inflammatory cytokines, mainly interleukins (IL-6 and IL8, IL1β), and Tumor Necrosis Factor(TNF)-α supported the view of endometriosis as an inflammatory disease. The concurrent prominent expressions of IL-10 an anti-inflammatory cytokine at the local endometriotic lesions and IL-2 needed in the stimulation and proliferation of regulatory T (Treg) cells indicated that the active priming of adaptive Tr1 regulatory cells takes place in endometriosis. The Treg priming probably started in the eutopic endometrium of the patients and continuing with enhanced strength in the disseminated endometriotic lesions. Moreover, the upregulation of mRNA for IL-10 and TGFβ1 without any upregulation of IL-2 in the peripheral blood mononuclear cells, further confirms that the Treg priming is a local phenomenon as a response to inflammation in endometriosis. The bias toward Treg cell priming is further proved by the abundance of Tregs in endometriotic tissue and peritoneal fluid of endometriosis patients.
Patients with endometriosis appear to have decreased cell-mediated immunity with suppressed T- and NK cell cytotoxicity. This can be explained by the altered immune response toward immunoregulation by adaptive Treg cells as a reaction to vigorous inflammation that may have an undesirable side effect by inhibiting the cytotoxic potency of the immune system in endometriosis. The regulatory T cells may be responsible for the suppression of immune surveillance at the endometriotic site since these cells can modify the secretion pattern of cytokines and suppress the activation and proliferation of immune cells like cytotoxic T and NK cells.
The study concluded that ectopic implantation in endometriosis is possibly a consequence of increased inflammation and priming of T regulatory cells that leads to impaired cytotoxicity and increased immune suppression.
Research Source: https://pubmed.ncbi.nlm.nih.gov/32623813/
Cytokine Immune response regulatory T cells cytotoxicity CD4 CD25 IL10 IL-8

