Menstural Blood: A Medium Full of Information for Reproductive Diseases
Apr 9, 2026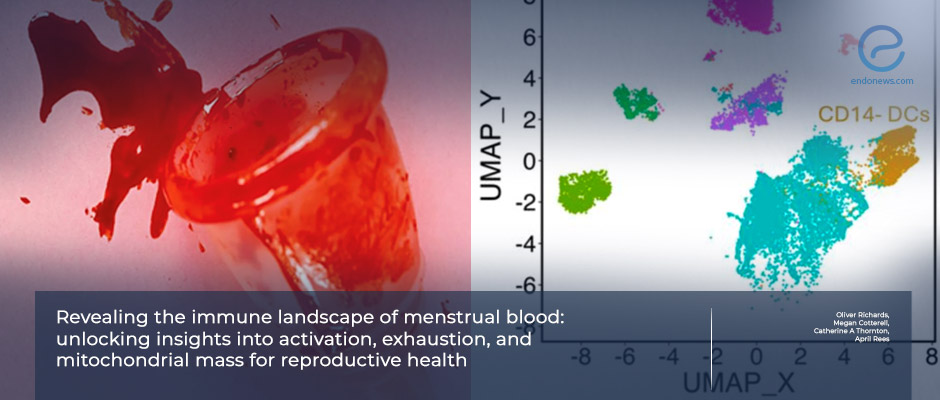
Menstrual blood could be a valuable tool to better understand the pathophysiology of reproductive diseases like endometriosis.
Key Points
Highlights:
- Menstrual blood harbors a distinct, tissue-adapted immune landscape differing fundamentally from peripheral blood.
- Disease-specific immune and metabolic patterns are observed in endometriosis and PCOS.
Importance:
- Menstrual blood represents a non-invasive, biologically relevant window into endometrial immune status.
- Enables immune profiling of reproductive diseases without invasive tissue sampling, supporting biomarker discovery and disease stratification.
What’s done here:
- This is an exploratory cross-sectional study of matched peripheral and menstrual blood from 23 women.
- 27-color flow cytometry performed in healthy controls (n=5), endometriosis (n=5), and PCOS (n=4).
- The immune cell profiles of mononuclear cells from the menstrual and peripheral blood were compared.
Key results:
- Menstrual mononuclear cells exhibit a distinct immune composition compared to Peripheral mononuclear cells: enriched in tissue-resident NK cells, macrophages, and dendritic cells.
- T cells in menstrual blood show increased mitochondrial mass, indicating a metabolically active, tissue-adapted phenotype.
- Immune checkpoint expression (e.g., PD-1) is elevated in specific T-cell subsets, suggesting activation/exhaustion dynamics.
- Endometriosis and PCOS demonstrate divergent, disease-specific immune signatures, rather than a uniform “reproductive disease” pattern.
- Distinct immune cell populations (e.g., dendritic cell subsets) may differentiate disease states.
Strengths and Limitations:
- Strengths are : first integration of immune and metabolic profiling in menstrual blood; high-dimensional, multi-parametric analysis capturing broad immune heterogeneity; direct comparison with matched peripheral blood.
- Limitations are: small exploratory cohort (n=4–5 per group); self-reported diagnosis without staging or hormonal context; lack of multiple comparison correction; findings are hypothesis-generating; mitochondrial mass used as a surrogate, not functional metabolic assessment.
From the Editor-in-Chief – EndoNews
Lay Summary
A study published in ImmunoHorizons by Dr. April Rees and colleagues from Swansea University Medical School, UK, demonstrates that menstrual blood provides a biologically informative and non-invasive window into immune processes underlying reproductive diseases.
Using an optimized high-dimensional,27-color flow cytometry platform incorporating mitochondrial profiling, the authors analyzed menstrual and matched peripheral blood mononuclear cells from healthy individuals and patients with endometriosis or polycystic ovary syndrome (PCOS).
This approach enabled detailed characterization of 40 immune cell subsets across 546 immunological parameters, including activation, migration, exhaustion, and metabolic features.
Menstrual blood exhibited a distinct immune landscape compared to peripheral blood, enriched in tissue-resident natural killer cells, macrophages, and dendritic cells, and characterized by increased mitochondrial mass in T-cell populations, consistent with a metabolically active, tissue-adapted immune environment.
Importantly, disease-specific immune signatures were observed. Endometriosis and PCOS displayed divergent patterns across multiple immune cell subsets, including regulatory T cells, CD4 central memory cells, plasmablasts, and dendritic cells, indicating that these conditions do not share a uniform immunological phenotype.
These findings position menstrual blood as a clinically accessible source for high-resolution immune profiling and support its potential role in biomarker discovery and disease stratification in reproductive medicine.
Research Source: https://pubmed.ncbi.nlm.nih.gov/41885004/
polycystic ovary syndrome menstrual blood biomarker

