A Possible genetic target for endometriosis treatment
Dec 3, 2020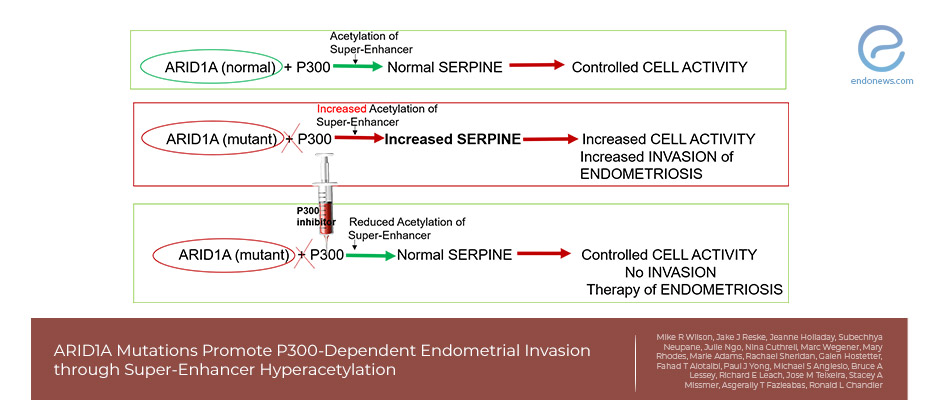
How to stop the spread of endometriosis?
Key Points
Highlight:
- Dr. Chandler's group found that invasive endometriosis, when the ARID1A gene is mutated, may be responsive to targeted therapies.
Background:
- Mutations in the SWI/SNF subunit ARID1A are frequent in endometriosis-related cancers and are also observed in deep infiltrating endometriosis, endometrioma, atypical endometrial hyperplasia, and endometrial cancer.
- ARID1A mutations may increase the risk of endometriosis and malignant transformation by providing a selective advantage to displaced endometrial cells.
Key points:
- In endometrial epithelium, ARID1A binding is associated with super-enhancers marked by high H3K27 acetylation, and loss of ARID1A leads to hyperacetylation and accessibility of this super-enhancer.
- ARID1A and P300 co-occupy highly active super-enhancers.
- P300 acetylates H3K27 and H3K18 that have known roles as super-enhancers, and P300 activity is required for invasion of an ARID1A mutant cell.
- Hyperacetylation of another super-enhancer, SERPINE1 also promotes ARID1A mutant-cell-invasion.
Conclusions:
- ARID1A prevention of super-enhancer hyperactivation plays an essential physiological role in maintaining endometrial tissue homeostasis and preventing cell invasion.
- Epigenetic dysregulation of super-enhancers promotes endometrial invasion and survival at ectopic sites.
- Alterations in super-enhancer activity may be an important feature of the endometriotic epithelium.
Lay Summary
Mutations in the chromosome remodeling complex SWI/SNF subunit ARID1A were first identified in ovarian clear-cell carcinoma and ovarian endometrioid carcinoma, two endometriosis-associated epithelial ovarian cancer subtypes. ARID1A gene mutations are also observed in deep ovarian and deep infiltrating endometriosis, atypical endometrial hyperplasia, and endometrial cancer. These mutations may increase the risk of endometriosis and malignant transformation by providing a selective advantage to displaced endometrial cells.
In this paper, Dr. Chandler's group identifies a mechanism by which ARID1A represses invasive phenotypes by antagonizing P300activity at super-enhancers. This paper was recently published in the journal named “Cell Reports”.
In endometrial epithelium, normally ARID1A binding through super-enhancers is marked by high H3K27 acetylation. The authors examined acetylation changes in ARID1A-deficient cells and observed decreased H3K27 acetylation following ARID1A loss. When the researchers tested and realized that ARID1A is strongly associated with P300, a histone acetyltransferase that acetylates H3K27 and H3K18 residues. Although P300 loss alone did not have any effect, ARID1A loss increased the cell invasion properties, and, co-knockdown of ARID1A and P300 completely rescued the invasion ARID1A mutant cells. demonstrating that P300 is required for the invasion properties of the ARID1A mutant cell.
ARID1A loss results in P300-dependent hyperacetylation and increased activity of the SERPINE1 super-enhancers increased SERPINE1 transcription for the acquisition of invasive phenotypes. SERPINE1 is a biomarker for endometriosis, with high levels of expression observed in ovarian and deep infiltrating endometriosis.
ARID1A prevention of super-enhancer hyperactivation plays an essential physiological role in maintaining endometrial tissue homeostasis and preventing cell invasion. This finding suggests that epigenetic dysregulation of super-enhancers promotes endometrial invasion and survival at ectopic sites. Hence, alterations in super-enhancer activity through blocking epigenetic pathways of P300 may be an important target to treat the invasion and spread of endometriosis epithelium. "Such a treatment may be a part of new management called “epigenetic therapy” would control the gene expression and could be far more effective than current treatments, including surgery, hormone therapy, and pain management", concluded the researchers.
Research Source: https://pubmed.ncbi.nlm.nih.gov/33176148/
Endometriosis ARID1A Super-Enhancer H3K27 P300 SERPINE1 epigenetic therapy endometriosis surgery hormone therapy

