Platelets Promote the Development of Endometriosis
May 30, 2017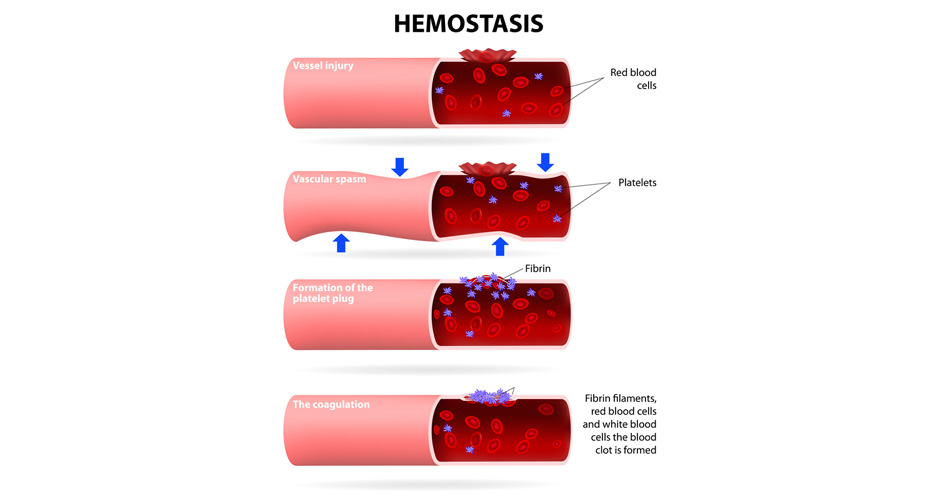
Blood clotting, tissue repair, and inflammation are important aspects that can all be related to platelets driven endometriosis pathogenesis. Since platelets can often release many factors during tissue repair processes, it is difficult to pin down exactly the mechanisms of how platelets could influence the reparative process of wound healing to maintain endometriosis.
Key Points
Highlight:
- Platelet cells can contribute to endometriosis pathogenesis.
Importance:
- If platelets contribute to endometriosis, anti-thrombotic therapy may be useful for management of endometriosis.
Whats done here:
- Platelet aggregation in 58 women with endometriosis and 47 without.
- Analysis of tissue factor in peritoneal fluid collected from endometriosis.
- Endometriosis animal model to examine the efficacy of platelet depletion.
Key Results:
- High platelet aggregation in endometriosis.
- Endometriosis peritoneal fluid had high tissue factor and induced platelets activation.
- Platelet depletion reduced endometriosis in an animal model.
Limitations:
- Cross-sectional study design, the results do not confer cause-effect relation, they show the coincidence.
- The study focused only on ovarian endometriomas, not other types of endometriosis.
Reference: Platelets are an unindicted culprit in the development of endometriosis: clinical and experimental evidence. Ding D, Liu X, Duan J, Guo SW. Hum Reprod. 2015 Apr;30(4):812-32. PMID: 25740881
Lay Summary
One hallmark of endometriosis is the recurrent or cyclical bleeding, which is indicative of vascular injury that often requires the wound healing or tissue repair system. To heal a wound, our body starts coagulation achieved by recruitment of activated platelet cells to the wound site where they adhere, aggregate and form clots to stop blood flow out of the tissue.
In a research article published in Human Reproduction (2015, vol. 30, page 812), Ding et al. hypothesized that platelets may be involved in the development of endometriosis because of the recurrent bleeding and healing processes. The study examined platelets aggregation in tissues from 58 women with endometriosis and 47 without.
Researchers found higher platelet aggregation in endometriosis than the control endometrium or the matched eutopic endometrium. Peritoneal fluid collected from the endometriosis patients induced a higher level of platelet activation, and a higher concentration of platelet tissue factor, important for the body’s blood coagulation cascades. Using anti-platelet antibody in endometriosis mice reduced >60% of the endometriosis size, while platelet infusion increased the endometriosis size by >30%. Furthermore, platelet depletion caused reduction of inflammatory mediators [such as Cyclooxygenase-2 (COX2)] and factors stimulating vasculogenesis and angiogenesis (such as vascular endothelial growth factor (VEGF)] and other wound healing and repair mediators [fibronectin, fibroblast growth factor receptor 2 (FGFR2)].
These data suggests that blood clotting, tissue repair, and inflammation are important aspects that can all be related to platelets driven endometriosis pathogenesis. Since platelets can often release many growth factors, cytokines, and chemokines during repair processes, it is difficult to pin down exactly the mechanisms of how platelets could influence the reparative process of wound healing to maintain endometriosis.
Nonetheless, this study is important and showed that platelets are involved in endometriosis development and creating a hypercoagulable state in women with endometriosis. With this knowledge, it can be implied that anti-thrombotic therapy may be useful for the treat of endometriosis. However, this hypothesis remains to be tested in preclinical models, as well as clinical trials.

